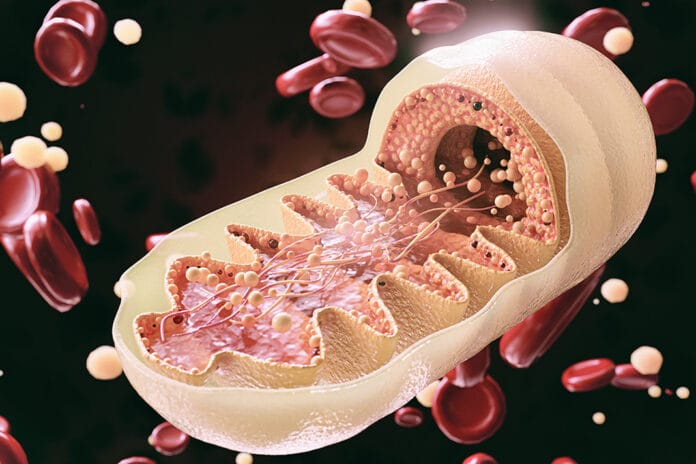
Mitochondria are the powerhouse of the cell, and we get all our mitochondria from our mothers (from the father in some rare cases). That is the extent of most people’s knowledge about mitochondria, which is really a shame because mitochondria are so much more than just the powerhouse of the cell.1
Mitochondrion (singular, as mitochondria is plural) contributes to cellular function and dysfunction, calcium signaling, cell growth and differentiation, and cell cycle control. They are essentially highly specialized bacteria. They are also a source of reactive oxygen species (ROS) and a central regulator of cell defense.
One of the most important functions of mitochondria, as it applies to dentistry, is that mitochondria regulate inflammation.5
Mitochondrial Disease
Let’s start by discussing mitochondrial disease. Mitochondrial disease is when the mitochondria can’t efficiently provide energy through the metabolism of sugar and oxygen. Essentially, the cells do not work correctly.2 Disease associated with mitochondrial disease includes fatigue, weakness, metabolic strokes, seizures, cardiomyopathy, arrhythmias, developmental or cognitive disabilities, diabetes mellitus, impairment of hearing, vision, growth, liver, gastrointestinal, or kidney function, and more.
Symptoms associated with mitochondrial disease can present at any age, from birth until late adulthood.3 Mitochondrial disease is genetic, most often inherited from the mother. As you can imagine, mitochondrial disease can have an impact on oral health.
Mitochondrial Dysfunction
Mitochondrial dysfunction differs slightly from mitochondrial disease. It can be an inherited mutation or be caused by environmental factors. It seems to be more relevant to dentistry since environmental factors such as bacteria associated with periodontal disease can be a contributing factor.4
Mitochondrial dysfunction is characterized by the reduction of high-energy molecules such as adenosine triphosphate (ATP) as well as loss of efficiency in the electron transport chain.6 This seems to be part of the aging process, but it also is evident in all chronic diseases. This list of diseases includes neurodegenerative disease (Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, etc.), cardiovascular diseases, diabetes and metabolic syndrome, autoimmune diseases, psychiatric diseases, gastrointestinal disorders, and periodontal disease.6,7
Tissues that require high energy production, such as the heart, brain, and liver, are areas that can be most affected by mitochondrial dysfunction. Though obesity isn’t a specific organ or tissue, it can contribute to mitochondrial dysfunction and the development of type 2 diabetes and metabolic syndrome as well as other chronic diseases associated with mitochondrial dysfunction. An excess of nutrients overwhelms the Krebs cycle (an important function of mitochondria in maintaining homeostasis within the cell), leading to increased production of ROS. The increased production of ROS causes oxidative stress and inflammation, which, if unchecked, can contribute to metabolic syndrome, insulin resistance, and type 2 diabetes.8
Mitochondrial Dysfunction Link to Periodontal Disease
A study published in the journal Cells identifies mitochondrial dysfunction, among other cellular processes, as an intracellular meeting point between diabetes mellitus and periodontal disease that might explain the link between the two diseases.9 The increased production of ROS when mitochondrial dysfunction is induced causes damage to the cellular components, impairing the inflammatory response. The impaired inflammatory response shifts the process into a pro-inflammatory state, influencing wound repair and modification of collagen structure, causing periodontal tissue to have a less reparative tendency and be more susceptible to periodontal breakdown.
Patients with type 2 diabetes and periodontal disease had higher levels of ROS when compared to healthy individuals. This is evident even when the individual only has one of the two diseases; however, it is more severe when both diseases are present in an individual. This assault can lead to mutations and/or polymorphisms in mitochondrial DNA. These mutations/polymorphisms contribute to poor metabolic control making management of diabetes and periodontal disease difficult.
A question that has yet to be answered is if environmentally induced mitochondrial dysfunction causes the disease or if the disease contributes to mitochondrial dysfunction. In the case of periodontal disease, in vitro studies showed P. gingivalis induced oxidative stress. This could cause damage to mitochondrial DNA and contribute to mitochondrial dysfunction. More studies are needed as these findings are very preliminary.10 Identifying the mechanism of which comes first will take time and may never be clearly identified. However, what is clear is that there is a connection between systemic diseases and periodontal disease via mitochondrial dysfunction.
Though there is still much to learn about mitochondrial dysfunction and how to prevent and treat it, the knowledge that it is a piece of the ever-growing puzzle that defines the oral-systemic link is of great value. Periodontal disease is a multifactorial disease that has bidirectional associations with multiple chronic systemic diseases. The more we know and understand about the development and progression and the factors linking periodontal disease to other systemic diseases, the closer we get to successfully managing and preventing periodontal disease.
Need CE? Click Here to Check Out the Self-Study CE Courses from Today’s RDH!
Listen to the Today’s RDH Dental Hygiene Podcast Below:
References
- McWilliams, T.G., Suomalainen, A. Mitochondrial DNA Can Be Inherited from Fathers, Not Just Mothers. Nature. 2019; 565(7739): 296-297. doi:10.1038/d41586-019-00093-1. Retrieved from https://www.nature.com/articles/d41586-019-00093-1
- Autism Spectrum Disorders: Frequently Asked Questions on Mitochondrial Disease. (n.d.). Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/ncbddd/autism/mitochondrial-faq.html
- Mitochondrial Disease. (n.d.). Children’s Hospital of Philadelphia. Retrieved from https://www.chop.edu/conditions-diseases/mitochondrial-disease
- Niyazov, D.M., Kahler, S.G., Frye, R.E. Primary Mitochondrial Disease and Secondary Mitochondrial Dysfunction: Importance of Distinction for Diagnosis and Treatment. Mol Syndromol. 2016; 7(3): 122-137. doi:10.1159/000446586. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4988248/
- Meyer, A., Laverny, G., Bernardi, L., et al. Mitochondria: An Organelle of Bacterial Origin Controlling Inflammation. Front Immunol. 2018; 9: 536. Published 2018 Apr 19. doi:10.3389/fimmu.2018.00536. Retrieved from https://www.frontiersin.org/articles/10.3389/fimmu.2018.00536/full
- Nicolson, G.L. Mitochondrial Dysfunction and Chronic Disease: Treatment with Natural Supplements. Integr Med (Encinitas). 2014; 13(4): 35-43. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566449/
- Govindaraj, P., Khan, N.A., Gopalakrishna, P., et al. Mitochondrial dysfunction and Genetic Heterogeneity in Chronic Periodontitis. Mitochondrion. 2011 May; 11(3): 504-12. doi: 10.1016/j.mito.2011.01.009. Epub 2011 Feb 2. PMID: 21296687. Retrieved from https://pubmed.ncbi.nlm.nih.gov/21296687/
- de Mello, A.H., Costa, A.B., Engel, J.D.G., Rezin, G.T. Mitochondrial Dysfunction in Obesity. Life Sci. 2018; 192: 26-32. doi:10.1016/j.lfs.2017.11.019. Retrieved from https://www.sciencedirect.com/science/article/pii/S092544392030185X?via%3Dihub
- Portes, J., Bullón, B., Quiles, J.L., et al. Diabetes Mellitus and Periodontitis Share Intracellular Disorders as the Main Meeting Point. Cells. 2021; 10(9): 2411. Published 2021 Sep 13. doi:10.3390/cells10092411. Retrieved from https://pubmed.ncbi.nlm.nih.gov/34572060/
- Bullon, P., Cordero, M.D., Quiles, J.L., et al. Mitochondrial Dysfunction Promoted by Porphyromonas gingivalis Lipopolysaccharide as a Possible Link between Cardiovascular Disease and Periodontitis. Free Radic Biol Med. 2011; 50(10): 1336-1343. Retrieved from https://pubmed.ncbi.nlm.nih.gov/21354301/