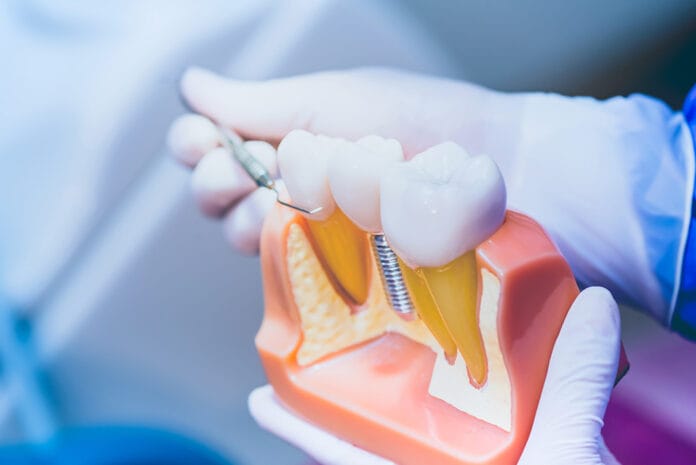
The concept of dental implants is not new. Evidence of dental implants has been dated all the way back to 600 AD and the Mayan population. While materials, techniques, and technologies have been improved upon, implants placed today and their predecessors both rely on the same process of osseointegration.1 Osseointegration is defined as “direct structural and functional connection between ordered, living bone and the surface of a load-carrying implant.”2 Osseointegration is considered imperative in the overall success of an implant.
The healing timeline of a dental implant will vary based upon many factors such as the type of implant used, if any additional surgical procedures (a sinus or ridge augmentation) need to be performed, and the quality of bone at the time of placement. The majority of dental implants will require a healing timeline of two to six months post-surgery before the restorative phase of treatment can begin.3
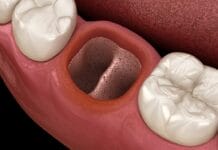
Peri-implant Health vs. Disease
The 2017 World Workshop hosted by the American Academy of Periodontology (AAP) and the European Federation of Periodontology (EFP) defined peri-implant health as having no clinical signs of inflammation and had the absence of bleeding upon probing (BOP).4 It is important to note that an implant can be considered “healthy” even if there is radiographic bone loss, as long as there is no inflammation present. Clinically, there is no parameter of probing depths that are considered normal or healthy for implants.4
The AAP has identified two types of peri-implant diseases: peri-implant mucositis and peri-implantitis. Clinically, peri-implant mucositis presents itself as inflammation around the implant coupled with BOP.4 Peri-implant mucositis is confined to the soft tissue around an implant only, and the supporting bone is not affected.5 Similar to its natural tooth counterpart, gingivitis, peri-implant mucositis is considered reversible.
Peri-implantitis will exhibit signs of inflammation and BOP similar to cases of peri-implant mucositis, but it will also display radiographic bone loss simultaneously.4 Due to the presence of bone loss, peri-implantitis is deemed irreversible. The presence or absence of bone loss, combined with inflammation and BOP, is critical in determining whether a case of peri-implant mucositis has progressed to peri-implantitis.
Contributing Risk Factors of Peri-implant Diseases
There are both local and systemic risk factors for developing peri-implant disease. Common local risk factors for peri-implant mucositis and implantitis include plaque, implant crown design, quality and quantity of gingival and mucosal tissue present, and residual/or excess cement.4
The following systemic risk factors play a role in an implant developing peri-implantitis (but does not apply to cases of peri-implant mucositis): Smoking, diabetes, heart disease, immunosuppression, history of periodontal disease, poor oral hygiene, periodontal disease, overloading of the implant, and previous implant failure.4,6
Smoking is, in fact, considered to be the largest systemic contributing factor to developing peri-implantitis. In these cases, there is a direct correlation between smoking and a reduced rate of osseointegration, which is detrimental to the long-term health of an implant.6
Diabetes has been shown to increase a patient’s risk for developing peri-implant disease, as it can hinder the body’s immune response and interfere with osteoblasts, bone regeneration, and wound healing.6
Previous periodontal disease is also considered a risk factor. It is believed that periodontal pathogens, specifically, Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, and Prevotella intermedia can recur, even years after periodontal therapy has been administered and cause peri-implantitis cases.6
As hygienists, we know that good oral hygiene lends itself to a healthy oral environment and is one factor that patients are directly in control of. Practicing good oral hygiene and following recommendations from their dental healthcare professionals in terms of recall intervals is recommended for all patient populations, but especially for those with implants since poor oral hygiene has been directly linked to peri-implantitis as well.6
One article stated that “The lack of regular prophylaxis resulted in a tendency for higher risk of developing peri-implantitis.”7
Restorative Risk Factors
The two main types of implant crowns are screw-retained and cement-retained. In regard to cement-retained restorations, residual cement that has been left behind after the restorative process is complete and is not entirely removed is considered a risk factor for peri-implantitis.7 One study evaluated 73 implants that had residual cement present found that 62 of those exhibited peri-implant disease.8
Residual cement can oftentimes be detected clinically as well as seen radiographically. An implant crown with an emergence profile greater than 30 degrees as well as a convex crown profile has also been shown to have a higher incidence of peri-implant disease.9
Nonsurgical Treatment
Nonsurgical treatment is typically reserved for peri-implant mucositis and some milder cases of implantitis. It should be noted that the majority of peri-implantitis ultimately needs surgical therapy even after nonsurgical therapy has been completed.7
Removal of bacteria pathogens from the implant surface is crucial in treating peri-implant disease. There continues to be contradictory information on which instruments and tools are the most efficient at achieving this while also causing the least amount of damage to the implant.
Stainless steel instruments are greatly recognized as too rough to be used on implant surfaces, while instruments designed specifically for implants that are made of softer materials like titanium or plastic have also been shown to damage the surface to some extent.7 However, the use of plastic instruments has been shown to leave residue behind on the implant surface which may lead to infection.10
Dental floss is also considered a controversial adjunctive homecare aid because floss remnants have been directly linked to cases of peri-implant disease.11
In regard to air polishing, one study states, “Research has indicated sodium bicarbonate based powders used with air polishing devices may become embedded in rough-surface implant surfaces.12 It has also been reported that water-soluble glycine powder used in specially designed air polishing devices has only small effects on implant surface topography.13 Another in vitro study assessed the effects of instrumentation on manufactured rough-surface implants and found that the metal instrumentation of these fixtures resulted in a smoother implant surface and less bacterial adherence.”14
The addition of laser therapy in conjunction with mechanical debridement has been proven to increase the resolution of peri-implantitis.7 While the use of systemic antibiotics to treat peri-implant disease remains controversial, local antibiotic use has been proven to help treat both peri-implant mucositis and implantitis.7
The clinician must weigh the risks versus benefits when determining the best instruments to use when treating peri-implant disease.
Surgical Treatment
In many cases, peri-implantitis is severe and can progress rapidly, so surgical treatment yields the best outcome. Most surgical procedures to treat implantitis include repositioning the tissue apically for better access, debriding the implant’s surface, which can include the use of hand instruments, ultrasonic, laser, or air abrasive tools, followed by the addition of bone grafting materials.7
The long-term success of surgical treatment can vary greatly depending upon the severity of the disease, patient compliance, and follow-up care from the treating dental professional.
The Hygienist’s Role
Because dental implants are considered predictable and are thought to have close to a 95% success rate, the number of implants hygienists will encounter in their careers will continue to rise.7
Peri-implant disease tends to be more aggressive and cause more damage in a shorter amount of time than periodontal disease in their natural teeth counterparts; hygienists must be vigilant in assessing implants for the presence of disease.
Developing an implant protocol that works best for everyone in the office may be beneficial. This may include exposing more frequent peri-apical radiographs, keeping implant-specific instruments and tools readily available, and keeping up to date and well-informed on the ever-changing research and data relating to both nonsurgical and surgical treatment modalities of peri-implant disease.
Before you leave, check out the Today’s RDH self-study CE courses. All courses are peer-reviewed and non-sponsored to focus solely on pure education. Click here now.
Listen to the Today’s RDH Dental Hygiene Podcast Below:
References
- Abraham, C.M. A Brief Historical Perspective on Dental Implants, Their Surface Coatings and Treatments. Open Dent J. 2014; 8: 50-55. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040928/
- Padmanabhan, T.V., Parithimarkalaignan, S. Osseointegration: An Update. J. Indian Prosthodont Soc. 2013; 13(1): 2–6. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3602536/
- Single Tooth Dental Implants. (n.d.). American Academy of Periodontology. Retrieved from https://www.perio.org/consumer/single-tooth-implant
- Armitage, G., Berglundh, T., Caton, J.G., Chapple, I.L.C., Jepsen, S., Kornman, K.S., Mealey, B.L., Papapanou, P.N., Sanz, M., Tonetti, MS. A new classification scheme for periodontal and peri‐implant diseases and conditions – Introduction and key changes from the 1999 classification. Journal of Periodontology. 2018; 89(S1): 1-8. Retrieved from https://aap.onlinelibrary.wiley.com/doi/10.1002/JPER.18-0157
- Peri-implant Diseases. (n.d.). American Academy of Periodontology. Retrieved from https://www.perio.org/consumer/peri-implant-disease
- Hammacher, C., Heiland, M., Henningsen, A., Smeets, R., Stein, J.M. Definition, etiology, prevention and treatment of peri-implantitis – a review. Head Face Med. 2014; 10: 34. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4164121/
- Alassy, H., Kormas, I., Pedercini, A., Pedercini, C., Raptopoulos, M., Wolff, L. Peri-Implant Diseases: Diagnosis, Clinical, Histological, Microbiological Characteristics and Treatment Strategies. A Narrative Review. Antibiotics (Basel). 2020; 9(11): 835. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7700146/
- Apse, P., Puisye, A., Linkeviciene, L., Linkeviciene, T., Vidasiute, E. Does residual cement around implant-supported restorations cause peri-implant disease? A retrospective case analysis. Clin Oral Implants Res. 2013; 24(11):1179-84. Retrieved from https://pubmed.ncbi.nlm.nih.gov/22882700/
- Chen, Y.W., Daub https://pubmed.ncbi.nlm.nih.gov/22882700/ert, D.M., Katafuchi, M., Leroux, B.G., Weinstein, B.F. Restoration contour is a risk indicator for peri‐implantitis: A cross‐sectional radiographic analysis. J Clin Periodontol. 2018; 45(2): 225-232. Retrieved from https://onlinelibrary.wiley.com/doi/abs/10.1111/jcpe.12829/
- Anand, B., Anand, V., Govila, V., Gulati, M. Implant Maintenance: A Clinical Update. Int Sch Res Notices. 2014; 2014: 6. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4897104/pdf/ISRN2014-908534.pdf
- Bruggenkate, M.M.T., Lang, N.P., Schulten, E., A., J., M., Velzen, F.J.J. V. Dental floss as a possible risk for the development of peri-implant disease: an observational study of 10 cases. Clin Oral Implants Res. 2016; 27(5):618-21. Retrieved from https://pubmed.ncbi.nlm.nih.gov/26261052/
- Chairay J.P., Boulekbache H., Jean A., Soyer A., Bouchard, P. Scanning electron microscopic evaluation of the effects of an air-abrasive system on dental implants: a comparative in vitro study between machined and plasma-sprayed titanium surfaces. J Periodontol. 1997; 68: 1215–1222. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6316998/
- Sahrmann P., Ronay V., Hofer D., Attin T., Jung R.E., Schmidlin, P.R. in vitro cleaning potential of three different implant debridement methods. Clin Oral Implants Res. 2015; 26:314–319. Retrieved from https://pubmed.ncbi.nlm.nih.gov/24373056/
- Duarte P.M., De Freitas, P.M., Ota-Tsuzuki C., Reis A.F. Bacterial adhesion on smooth and rough titanium surfaces after treatment with different instruments. J Periodontol. 2009; 80: 1824–1832. Retrieved from https://pubmed.ncbi.nlm.nih.gov/19905952/