According to the Centers for Disease Control and Prevention (CDC, 2019), approximately 60% to 70% of women experience gingivitis and early periodontitis during pregnancy. Periodontal disease is a common inflammatory disease consisting of two components, gingivitis and periodontitis.
Gingivitis is inflammation of the soft tissue surrounding the teeth and gingiva. Periodontitis is the inflammatory reaction responsible for the destruction of the periodontal ligaments, alveolar bone, cementum, and soft tissues. Multiple gram-negative anaerobic bacteria are responsible for the destructive nature of periodontitis. These bacterial species include Porphyromonas gingivalis, Fusobacterium nucleatum, Prevetello intermedia, Actinobacillus actinomycetemcomitans, and Treponema denticola and are commonly found in the gingival sulcus.
Periodontal disease is a known risk factor for several systemic diseases, including pneumonia (in older adults), diabetes mellitus, and atherosclerosis (Komine-Aizawa et al., 2019). Research has examined a correlation between maternal periodontal disease and adverse birth outcomes (ABO), which include preterm birth, low birth weight, the early loss of a pregnancy, and preeclampsia (Xiong et al., 2006).
The purpose of this paper is to discuss the literature surrounding the correlation between maternal periodontal disease and adverse birth outcomes.
Epidemiology
The World Health Organization (WHO, 2019) estimates 15 million preterm infants are born each year. Preterm birth is defined as birth of an infant before 37 weeks gestation. Approximately 1 million infant deaths each year are due to preterm delivery, and it is the leading cause of death in children under five years of age (WHO, 2019).
Infants who survive preterm birth can have lifelong medical conditions such as respiratory issues, feeding difficulties, cerebral palsy, learning disabilities, and visual and hearing problems (March of Dimes, 2019). Risk factors for preterm birth include previous delivery of a premature infant, pregnancy with multiples, infection or chronic condition (diabetes or high blood pressure), cigarette smoking, alcohol use, use of street drugs, maternal weight, stressful life events (death of loved one or domestic violence), multiple miscarriages or abortions, and physical trauma (Mayo Clinic, 2019).
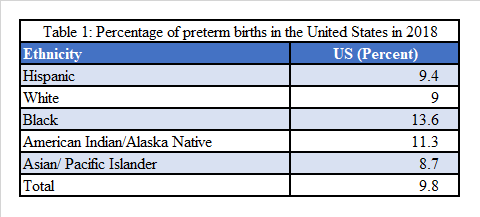
Statistics show that in the United States, approximately 14% of preterm births were to African American women in 2018, equating to one in seven births (March of Dimes, 2018). White non-Hispanic women accounted for 9% of the preterm births in the United States. (See Table 1)
Low birth weight (LBW) is defined as birth weight less than 2,500 grams, or five pounds and five ounces. This condition is commonly associated with preterm births. Infants born with LBW have difficulty eating, gaining weight, and fighting infection (March of Dimes, 2019). Long-term health conditions associated with LBW include diabetes, heart disease, high blood pressure, lung and kidney issues, developmental delays, and obesity (March of Dimes, 2019).
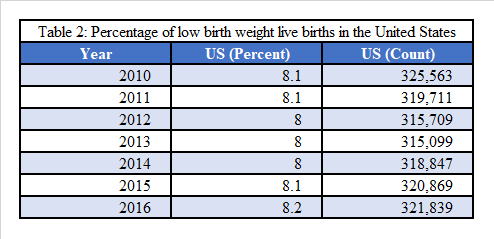
A noteworthy risk factor that contributes to LBW is race/ethnicity, as black non-Hispanic women are two times more likely to have an infant born with LBW than white non-Hispanic women (Children’s Hospital of Philadelphia, 2014). Other risk factors include maternal age (women <17 and >35 are at highest risk), socioeconomic status, poor nutrition, inadequate prenatal care, and illicit use of drug, alcohol, and cigarettes (March of Dimes, 2016). Over the last decade, the United States observed a decrease, then a rise in LBW births, displayed in Table 2 (March of Dimes, 2016). In 2016, nearly 322,000 live births were considered LBW.
The reduction or elimination of possible risk factors that may contribute to the risk of an adverse birth outcome, specifically LBW and preterm births, is necessary to reduce morbidity. Risk factors include the possible association between ABO and periodontal disease. Collaborating with obstetricians to educate pregnant women on the importance and safety of oral care before, during, and after pregnancy would help to decrease these risk factors.
Determinants
Periodontal disease as a risk factor for ABO is not a new concept. In 1916, Galloway identified infection of the teeth, tonsils, sinuses, and kidneys could affect the developing fetus (Jared & Boggess, 2008). Pregnancy brings on both physiological and hormonal changes. These changes cause an increase in the inflammatory response, which can increase the risks of developing periodontal disease (Jared & Boggess, 2008).
The prevalence of pregnancy gingivitis can vary from 36% to 100%, depending on the research conducted (Kierce et al., 2017). During the second and eighth months of pregnancy, the surge of progesterone and estrogen affects the blood vessels of the gingiva by increasing permeability. The permeability allows bacteria to enter the bloodstream and potentially reach the fetal-placenta unit, destroying the placental walls. Pathogenic destruction of the placenta causes a breakdown in the tissue that can cause preterm labor and LBW (Komine-Aizawa et al., 2019).
Analysis
Several studies have aimed to evaluate the relationship between periodontal disease and ABO. Komine-Aizawa et al. (2019), concluded there were two mechanisms for how periodontal bacteria entered the maternal reproductive system. The direct pathway was the dissemination of pathogenic bacteria through the bloodstream creating a secondary inflammatory response (Komine-Aizawa et al., 2019).
Indirectly, the inflammatory cytokines reach the placenta, creating a secondary infection in the fetal-placental unit (Komine-Aizawa et al., 2019). Similar to the destruction of the oral tissues due to inflammatory cytokines, the production of inflammatory cytokines in the fetal-placental interface destroys the placental tissue, affecting the structural integrity of the placenta (Komine-Aizawa et al., 2019). Destruction of the placental walls causes the body to respond with uterine contractions, therefore causing preterm labor. Inflammatory cytokines have often been found in the amniotic fluid of preterm births (Kierce et al., 2017).
Between 2002 and 2016, seven studies concluded a significant relationship between active periodontal disease in pregnant women and ABO (Kierce et al., 2017). Corbella et al. (2016) reviewed 22 different cohort and case-control studies, concluding a weak association between periodontal disease and ABOs. Though the correlation was low, it reiterates the importance of additional future research to bring awareness to the relationship. Furthermore, these findings accentuate the necessity of promoting adequate oral care before, during, and after pregnancy.
Although Corbella et al. (2016) showed promising results, other studies reveal contradictory findings. Vettore et al. (2008) concluded there was little influence of periodontal disease on low preterm birth weight (PTLBW) (Vettore et al., 2008). Ali and Abiden (2012) concluded an increase in poor periodontal health did not result in ABO. The number of participants, demographic, and difference in the study definitions are some discrepancies in this research.
Other studies were performed to determine if the treatment of periodontal disease during pregnancy would result in a lower risk of ABO. Jeffcoat et al. (2011), Lopez et al. (2005), Offenbacher et al. (2001), and Offenbacher et al. (2006) concluded the treatment of severe periodontitis in pregnant women resulted in a reduction in the prevalence of preterm births.
Studies conducted by Fiorini et al. (2013) and Kaur et al. (2014) established treatment of maternal periodontal disease improved oral conditions and decreased the inflammatory receptors. However, treatment did not lower the risk of ABO. Lastly, studies conducted by Han et al. (2011), Horton et al. (2012), Michalowicz et al. (2006), and Xiong et al. (2011) resulted in little to no positive outcome of periodontal treatment during pregnancy and the reductions of ABO.
In the literature, many studies focused solely on periodontal health and birth outcomes. Other determining factors such as maternal age, race, and use of cigarettes, alcohol, and illicit drugs were not part of the collected data.
Conclusion
Currently, the evidence does not support a direct causal relationship between maternal periodontal disease and ABO. However, the research does reveal a possible correlation as well as the benefits of treating periodontal disease during pregnancy.
Health-care professionals agree inflammation in the oral cavity can cause an inflammatory response in other areas of the body. Establishing protocols for the treatment of periodontal disease can decrease the host response. In 2013, the American College of Obstetricians and Gynecologists and the American Academy of Periodontology presented documentation regarding the necessity and importance of oral care during pregnancy.
Oral health professionals and OBGYNs should take the time to discuss the risks of ABO and the importance of good oral hygiene habits during pregnancy. Although there is still a need for more research regarding periodontal health and ABOs, prevention is the best way to remove periodontal disease as a factor.
Before you leave, check out the Today’s RDH self-study CE courses. All courses are peer-reviewed and non-sponsored to focus solely on high-quality education. Click here now.
Listen to the Today’s RDH Dental Hygiene Podcast Below:
References
- Ali, T.B., Abiden, K.Z. Relationship of periodontal disease to pre-term low birth weight infants in a selected population – a prospective study. Community Dental Health. 2012; 29(1): 100-105. Retrieved from https://www-cdhjournal-org.ezproxymcp.flo.org/issues
- American College of Obstetricians and Gynecologists. (2013). Oral health care during pregnancy and through the lifespan. Retrieved from https://www.acog.org/Clinical-Guidance-and-Publications/Committee-Opinions/Committee-on-Health-Care-for-Underserved-Women/Oral-Health-Care-During-Pregnancy-and-Through-the-Lifespan?IsMobileSet=false
- Center for Disease Control and Prevention. (2019, February 19). Pregnancy and oral health. Retrieved from https://www.cdc.gov/oralhealth/publications/features/pregnancy-and-oral-health.html
- Children’s Hospital of Philadelphia (2014, August 23). Low Birthweight. Retrieved from https://www.chop.edu/conditions-diseases/low-birthweight
- Corbella, S., Taschieri, S., Del Fabbro, M., Francetti, L., Weinstein, R., & Ferrazzi, E. (2016). Adverse pregnancy outcomes and periodontitis: A systematic review and meta-analysis exploring potential association. Quintessence International (Berlin, Germany: 1985). 2016; 47(3): 193–204. Retrieved from https://qi.quintessenz.de/index.php?doc=abstract&abstractID=34980
- Fiorini, T., Susin, C., da Rocha, J. M., Weidlich, P., Vianna, P., Moreira, C. H. C.,… Oppermann, R. V. (2013). Effect of nonsurgical periodontal therapy on serum and gingival crevicular fluid cytokine levels during pregnancy and postpartum. Journal of Periodontal Research. 2013; 48(1): 126–133. Retrieved from https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1600-0765.2012.01513.x
- Han, Y.W. (2011) Oral health and adverse pregnancy outcomes – what’s next? Journal of Dental Research. 2011; 90(3): 289-293. Retrieved from https://doiorg.ezproxymcp.flo.org/10.1177/0022034510381905
- Horton, A.L., Boggess, K.A. (2012). Periodontal disease and preterm birth. Obstetrics and Gynecology Clinics of North America. 2012; 39(1); 17-23, vii. Retrieved from https://www.sciencedirect.com/science/article/abs/pii/S0889854511001318?via%3Dihub
- Jared, H., Boggess, K. A. (2008). Periodontal diseases and adverse pregnancy outcomes: A review of the evidence and implications for clinical practice. American Dental Hygienists’ Association. 2008; 82(suppl 1): 24–24. Retrieved from https://jdh.adha.org/content/82/suppl_1/24.abstract
- Jeffcoat, M., Parry, S., Sammel, M., Clothier, B., Catlin, A., Macones, G. (2011). Periodontal infection and preterm birth: Successful periodontal therapy reduces the risk of preterm birth. BJOG: An International Journal of Obstetrics and Gynecology. 2011; 118(2): 250-256. Retrieved from https://obgyn.onlinelibrary.wiley.com/doi/full/10.1111/j.1471-0528.2010.02713.x
- Kaur, M., Geisinger, M. L., Geurs, N. C., Griffin, R., Vassilopoulos, P. J., Vermeulen, L., Reddy, M. S. (2014). Effect of intensive oral hygiene regimen during pregnancy on periodontal health, cytokine levels, and pregnancy outcomes: A pilot study. Journal of Periodontology. 2014; 85(12): 1684–1692. Retrieved from https://aap.onlinelibrary.wiley.com/doi/abs/10.1902/jop.2014.140248
- Kierce, E. & Rainchuso, L. (2017). Oral health’s involvement in adverse birth outcomes. Retrieved from https://decisionsindentistry.com/article/oral-healths-involvement-adverse-birth-outcomes/
- Komine-Aizawa, S., Aizawa, S., & Hayakawa, S. (2019). Periodontal diseases and adverse
- pregnancy outcomes. The Journal of Obstetrics and Gynecology Research. 2019; 45(1): 5–12. Retrieved from https://obgyn.onlinelibrary.wiley.com/doi/full/10.1111/jog.13782
- Lopez, N.J., Da Silva, I., Ipinza, J., Gutierrez, J. (2005). Periodontal therapy reduces the rate of preterm low birth weight in women with pregnancy-associated gingivitis. Journal of Periodontology. 2005; 76(11 Suppl): 2144-2153. Retrieved from https://aap.onlinelibrary.wiley.com/doi/abs/10.1902/jop.2005.76.11-S.2144
- March of Dimes. (n.d.-a). Low birthweight United States | PeriStats. Retrieved from https://www.marchofdimes.org/peristats/ViewTopic.aspx?reg=99&top=4&lev=0&slev=1
- March Of Dimes. (n.d.-b). Preterm by race/ethnicity United States | PeriStats. Retrieved from https://www.marchofdimes.org/Peristats/ViewSubtopic.aspx?reg=99&top=3&stop=63&lev=1&slev=1&obj=1
- March of Dimes. (2018, March). Low birthweight. Retrieved from https://www.marchofdimes.org/complications/low-birthweight.aspx
- March of Dimes (2019, October). Premature babies. Retrieved from https://www.marchofdimes.org/complications/premature-babies.aspx
- Mayo Clinic. (2019). Premature birth—Symptoms and causes. Retrieved from https://www.mayoclinic.org/diseases-conditions/premature-birth/symptoms-causes/syc-20376730
- Offenbacher S, Lieff S, Boggess KA, et al. (2001). Maternal periodontitis and prematurity. Part I: Obstetric outcome of prematurity and growth restriction. Annals Periodontology. 2001; 6(1): 164-174. Retrieved from https://aap.onlinelibrary.wiley.com/doi/abs/10.1902/annals.2001.6.1.164
- Offenbacher S, Lin D, Strauss R, et al. (2006). Effects of periodontal therapy during pregnancy on periodontal status, biologic parameters, and pregnancy outcomes: A pilot study. Journal of Periodontology. 2006; 77(12): 2011-2024. Retrieved from https://aap.onlinelibrary.wiley.com/doi/abs/10.1902/jop.2006.060047
- Tettamanti, L., Lauritano, D., Narbone, M., Gargari, M., Silvestre-Rangil, J., Gavoglio, P., & Tagliabue, A. (2017). Pregnancy and periodontal disease: Does exist a two-way relationship? Oral & Implantology, 10(2), 112–118. https://doi.org/10.11138/orl/2017.10.2.112
- Vettore, M. V., Leal, M. doC., Leão, A. T., da Silva, A. M. M., Lamarca, G. A., & Sheiham, A. (2008). The Relationship between Periodontitis and Preterm Low Birthweight. Journal of Dental Research. 2008; 87(1): 73–78. Retrieved from https://journals.sagepub.com/doi/10.1177/154405910808700113
- Xiong, X., Buekens, P., Fraser, W. D., Beck, J., & Offenbacher, S. (2006). Periodontal disease and adverse pregnancy outcomes: A systematic review. BJOG: An International Journal of Obstetrics and Gynecology. 2006; 113(2):135-43. Retrieved from https://obgyn.onlinelibrary.wiley.com/doi/full/10.1111/j.1471-0528.2005.00827.x
- Xiong X, Buekens P, Goldenberg RL, Offenbacher S, Qian X. (2011). Optimal timing of periodontal disease treatment for prevention of adverse pregnancy outcomes: Before or during pregnancy? American Journal of Obstetrics and Gynecology. 2011; 205(2): 111.e1-111.e6. Retrieved from https://www.ajog.org/article/S0002-9378(11)00326-7/fulltext
- World Health Organization. (2019). Preterm birth. Retrieved from https://www.who.int/news-room/fact-sheets/detail/preterm-birth