Are you in need of CE credits? If so, check out our peer-reviewed, self-study CE courses here.
Test Your Infection Control Knowledge
1. There are no documented cases of the transmission of infectious agents involving patients or dental health care personnel in a dental setting.
Despite the rarity of the transmission of infectious agents in dental settings, cases involving patients and dental health care personnel (DHCP), including patient-to-patient transmission, have been documented, indicating a persistent risk. In most cases, investigators are unable to identify a specific infection prevention failure for every transmission. However, identified breakdowns have involved unsafe injection practices, neglecting to heat-sterilize equipment between patients, and failing to monitor autoclaves (e.g., spore testing). These incidents suggest a need for comprehensive training to equip DHCP with a full understanding of the principles, recommended practices, and implementation of infection control, as well as the specific conditions that allow infectious diseases to spread.1
Reference
1. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. (2016, October). Centers for Disease Control and Prevention: U.S. Department of Health and Human Services. https://www.cdc.gov/dental-infection-control/media/pdfs/2024/07/safe-care2.pdf
2. Every dental setting should have at least one infection prevention coordinator. This individual is responsible for written policies that are strictly limited to the Occupational Safety and Health Administration (OSHA) bloodborne pathogens standard.
Infection prevention is a priority for all dental settings. This requires a structured administrative approach that goes beyond compliance with the OSHA bloodborne pathogen standard to provide comprehensive patient safety. To make sure infection prevention measures are implemented, at least one trained infection prevention coordinator is responsible for developing, maintaining, and overseeing a formal infection prevention program.1
The infection prevention coordinator(s) should create written policies and procedures based on evidence-based guidelines, regulations, or standards. These policies and procedures should be tailored to the specific dental setting and the patient population served. Additionally, these protocols should be reassessed at least annually or according to state or federal requirements.1
Reference
1. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. (2016, October). Centers for Disease Control and Prevention: U.S. Department of Health and Human Services. https://www.cdc.gov/dental-infection-control/media/pdfs/2024/07/safe-care2.pdf
3. Which of the following terms describes the minimum infection prevention practices that should be followed in a dental setting?
The Centers for Disease Control and Prevention recommended the term and use of Standard Precautions in 1996.1 Standard Precautions are the minimum infection prevention practices that apply to all patients, regardless of a patient's known or suspected infection status.1,2 These protocols serve a dual purpose: protecting DHCP from occupational exposure to infectious agents and preventing DHCP from spreading infections among patients.2
The core components include:
- Hand hygiene
- Use of personal protective equipment (PPE)
- Respiratory hygiene/cough etiquette
- Sharps safety
- Safe injection practices
- Sterile instruments and devices
- Clean and disinfected environmental surfaces2
Because the effectiveness of these measures relies on informed decision-making, ongoing education and training are considered critical for ensuring DHCP compliance.2
References
1. OSHA Healthcare Standards. (2022, April 28). Centers for Disease Control and Prevention: National Institute for Occupational Safety and Health. https://www.cdc.gov/niosh/learning/safetyculturehc/module-5/5.html
2. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. (2016, October). Centers for Disease Control and Prevention: U.S. Department of Health and Human Services. https://www.cdc.gov/dental-infection-control/media/pdfs/2024/07/safe-care2.pdf
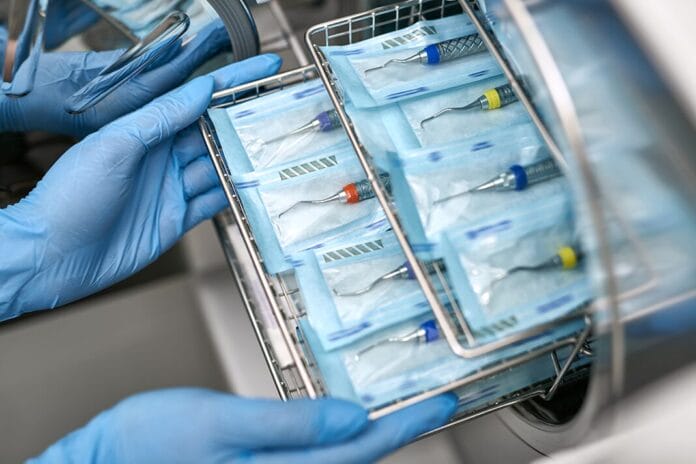
4. Handpieces that connect to the air- and waterlines of dental units (corded devices) should always be cleaned and heat-sterilized between patients.
Handpieces that connect to the air- and waterlines of dental units (corded devices) should always be cleaned and heat-sterilized between patients, according to the manufacturer's instructions for use. Although considered semicritical items, research has shown that their internal components can become contaminated with patient material during use. Without proper cleaning and heat sterilization, the next patient could be exposed to potentially infectious materials.1,2
Immersion in high-level disinfectants or surface disinfection (simply wiping the surface) are not acceptable methods for reprocessing handpieces.1,2 If a handpiece lacks U.S. Food and Drug Administration (FDA) clearance with validated manufacturer instructions for reprocessing and cannot be heat-sterilized, it should not be used.2
For handpieces that do not connect to the air- and waterlines of dental units (cordless devices), DHCP should use only FDA-cleared devices and adhere to the manufacturer's instructions for use regarding cleaning and sterilization.2
References
1. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. (2016, October). Centers for Disease Control and Prevention: U.S. Department of Health and Human Services. https://www.cdc.gov/dental-infection-control/media/pdfs/2024/07/safe-care2.pdf
2. Best Practices for Dental Handpieces. (2024, May 15). Centers for Disease Control and Prevention. https://www.cdc.gov/dental-infection-control/hcp/dental-ipc-faqs/dental-handpieces.html
5. When reprocessing instruments, they do not need to be dry before wrapping or bagging. To confirm effective sterilization, dental practices should use a single-monitoring system, choosing either mechanical, chemical, or biological monitoring.
The reprocessing cycle begins with cleaning using automated equipment, such as an ultrasonic cleaner or a washer disinfector, to minimize exposure to blood and remove organic debris that could otherwise shield microbes, potentially compromising sterilization. Once cleaned and dried, instruments should be inspected, wrapped or bagged, and labeled with the date, load number, and sterilizer used before undergoing heat sterilization.1
To confirm the sterilization process is effective, a triple-monitoring system should be used:
- Mechanical: Check sterilizer gauges and displays, and document pressure, temperature, and exposure time.
- Chemical: Chemical indicator tapes, strips, or special markings on packaging materials indicate that sterilization conditions have been met (e.g., time, temperature, and/or the presence of steam). An indicator strip should be used inside every package to verify heat or steam penetration.
- Biological: Spore testing should be performed at least weekly to confirm the actual destruction of resistant microorganisms.1
Following the manufacturer's instructions when using mechanical, chemical, and biological monitors helps ensure the effectiveness of the sterilization process. Accurate sterilization monitoring and equipment maintenance records should be maintained according to state and local regulatory requirements. This also helps ensure that sterilization parameters have been met and indicates if there's a problem.1
Reference
1. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. (2016, October). Centers for Disease Control and Prevention: U.S. Department of Health and Human Services. https://www.cdc.gov/dental-infection-control/media/pdfs/2024/07/safe-care2.pdf