One of the most important aspects of an appointment is reviewing health histories with patients. Every dental hygienist has experienced that moment, where one checked box sends our minds searching for the right questions to determine if it is safe to proceed with treatment for both the patient and ourselves.
Tuberculosis tends to be one of those recurring conversation topics where some dental hygienists are still unsure about how to proceed when a patient living with active or latent tuberculosis presents in our office, so let’s refresh our knowledge!
What is Tuberculosis?
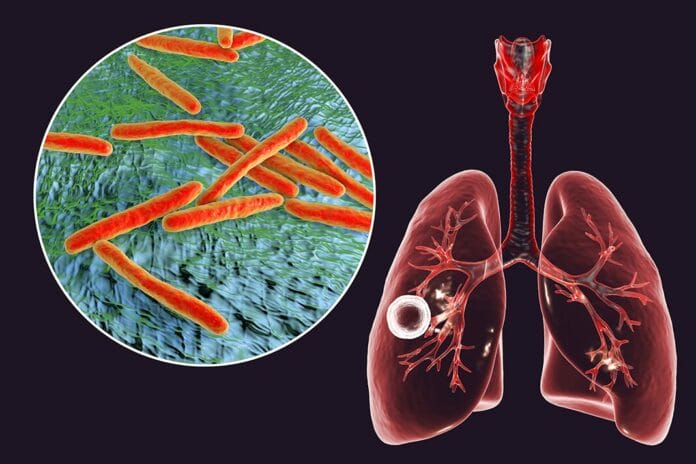
Tuberculosis (TB) is caused by a bacterium called Mycobacterium tuberculosis that most often attacks lung alveoli but may also affect other parts of the body like the spine, kidneys, and brain.2 Tuberculosis has existed throughout human history, the causative bacterium was identified by Robert Koch early in the 19th century, and airborne transmission was later proven in the 1950s.6 Since not everyone who is infected by M. tuberculosis becomes ill, there are two TB-related conditions: TB disease and latent tuberculosis infection (LTBI).
Those with active tuberculosis may display symptoms such as a cough lasting more than three weeks that produces phlegm or blood, chest pain, fatigue, fever, chills, night sweats, and unexplained weight loss.
Tuberculosis can be detected by a positive tuberculosis skin test (TST) and interferon gamma release assay (IGRA) blood test. These tests indicate the presence of infection by M. tuberculosis but cannot differentiate from the active or latent disease. In order to confirm active tuberculosis, a chest x-ray and sputum samples are needed. Active tuberculosis has a much higher burden of TB bacilli than LTBI.2,6
TB can live in the body without causing illness, and those with latent tuberculosis infection may have a positive TST or IGRA but do not feel sick at all. People with latent tuberculosis infection generally have no clinical symptoms, radiological abnormalities, or microbiological evidence of TB. Without proper treatment, there is a 5-10% chance of LTBI becoming active TB disease with the highest risk in the first two years after the initial infection.3,4 It should be emphasized here that a person with latent tuberculosis infection cannot spread tuberculosis.1,2
How is Tuberculosis Spread?
Tuberculosis is spread when someone with active TB disease of the lungs or throat coughs, speaks or sings. TB is not spread by kissing, sharing a toothbrush or bed linens, shaking hands, touching toilet seats, or sharing food and drink.2 Transmission is influenced by the level of host immunoresistance, the infectiousness of virulent TB bacilli, the strength of the infectious source, environmental stresses, and biosocial factors. Droplet nuclei measuring 1-5µm and containing 1-10 TB bacilli can remain in the air for several hours and are inhaled into lungs and alveoli.1,6 Exposure risk increases depending on the concentration of droplet nuclei in the air and duration of exposure to an infected person.4
How is Tuberculosis Treated?
Tuberculosis first-line medications include isoniazid (INH), rifampin (RIF), ethambutol (EMB), and pyrazinamide (PZA). The CDC recommends regimens of a combination of these drugs for 6-9 months for active tuberculosis. It is also important for the patient to be as isolated as possible until tests are clear of infection.
For the treatment of latent tuberculosis infection, the CDC recommends once weekly isoniazid-rifapentine for twelve weeks. In both cases, it is extremely important for patients to both take the medication correctly and finish it completely to avoid risking recurrence or becoming resistant to drug therapy.2
Multi-drug resistant tuberculosis (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB) have emerged due to inappropriate use of TB medications, improper prescribing, poor drug quality, and patients stopping treatment prematurely.5 In response, WHO began recommending the use of Xpert MTB/RIF® in 2010 which can simultaneously detect tuberculosis and resistance to rifampicin.8 Directly observed therapy short course programs (DOTS) conducted in conjunction with a regular visit from a healthcare provider have also been recommended by WHO to ensure patient compliance and reduce drug resistance.5
The Bacille Calmette-Guerin (BCG) vaccine was first used in humans in 1921. The vaccine is mainly used in developing countries where the incidence of tuberculosis is very high and is also given to infants who will be in the care of those with LTBI.5 The vaccine may cause a positive tuberculosis skin test, but the interferon-gamma release assay blood test will not give a false-positive.2
What Are The Oral Manifestations of Tuberculosis?
Oral lesions associated with tuberculosis may be an important step in early diagnosis and treatment. However, differential diagnosis of lesions associated with tuberculosis is extremely difficult, especially before systemic symptoms have presented.5 TB bacilli can spread through the bloodstream to other parts of the body including the maxilla and mandible. Only 0.5-1% of tuberculosis patients report oral lesions and are rare to encounter.
Primary lesions are usually painless ulcers with regional lymph node enlargement, while secondary lesions can also be singular but indurated, painful, irregular ulcers covered by inflammatory exudates.
Primary lesions are more often seen in children, and secondary lesions more-so in middle-aged and older adults. The most common area for tuberculosis-related oral lesions is the tongue, and treatment is the same as systemic tuberculosis medication regimens.5
It is thought that the oral cavity’s resistance to tuberculosis can be attributed to saliva, saprophyte presence, the resistance of striated muscles to bacterial invasion, and the thickness of protective epithelial coverings.5
Infection Control Precautions for the Dental Office
The purpose of a dental infection-control program is to provide a safe working environment that reduces the risk of both healthcare-associated infections among patients and occupational exposures among dental team members.5 Each office should assign responsibility for TB infection control in their office, complete a risk assessment, create a written infection-control plan, and the entire staff should be trained and educated on TB prevention, transmission, and symptoms.3
Dental hygienists should be aware of the signs and symptoms of tuberculosis, onset, duration, risk factors, past exposures, test results, and if LBTI treatment was completed. If you are concerned a patient may have active tuberculosis they should be removed from any common areas with other patients and staff, given a mask, assessed for the urgency of dental needs, and referred immediately for medical care.1
Urgent dental care of a patient with active TB should only be provided in an OSHA standard respiratory airborne infection isolation room, and providers should be fitted with N-95 respirators since surgical masks do not filter out TB bacilli.
Latent tuberculosis is not infectious, and standard infection control precautions are sufficient for these patients.1 Drills, ultrasonic instruments, and anything else that may create an aerosol is only contraindicated with active TB disease.
When Should Dental Hygienists be Tested for Tuberculosis?
Recent studies have brought into question the cost-effectiveness of serial occupational testing since TB incidence rates among healthcare workers have been shown to be similar to the general population.7 The ADA recommends that all dental staff have a baseline TST or IGRA at the beginning of employment. Annual testing is recommended for dental hygienists who work in a medium-risk setting but not for those in low-risk settings (defined as a practice seeing less than three TB patients per year) unless exposed.1,2 Table 1 outlines the most recent updates to CDC recommendations.
| Category | 2005 Recommendation | 2019 Recommendation |
| Baseline (preplacement) screening and testing | TB screening of all HCP, including a symptom evaluation and test (IGRA or TST) for those without documented prior TB disease or LBTI. | TB screening of all HCP, including a symptom evaluation and test (IGRA or TST) for those without documented prior TB disease or LTBI (unchanged); individual TB risk assessment (new). |
| Postexposure screening and testing | Symptom evaluation for all HCP when exposure is recognized. For HCP with a baseline negative TB test and no prior TB disease or LTBI, perform a test (IGRA or TST) when the exposure is identified. If that test is negative, do another test 8-10 weeks after last exposure. | Symptom evaluation for all HCP when exposure is recognized. For HCP with a baseline negative TB test and no prior TB disease or LTBI, perform a test (IGRA or TST) when the exposure is identified. If that test is negative, do another test 8-10 weeks after last exposure. (unchanged). |
| Serial screening and testing for HCP without LTBI | According to health care facility and setting risk assessment. Not recommended for HCP working in low-risk health care settings and settings with potential ongoing transmission. | Not routinely recommended (new); can consider for selected HCP groups (unchanged); recommended annual TB education for all HCP (unchanged), including information about TB exposure risks (new emphasis). |
| Evaluation and treatment of positive test results | Referral to determine whether LBTI treatment is indicated. | Treatment is encouraged for all HCP with LBTI, unless medically contraindicated (new). |
Table 1: Comparison of 2005 recommendations versus 2019 recommendations. Adapted from Lee, S.H. (2016). Tuberculosis Infection and Latent Tuberculosis. Tuberculosis and respiratory diseases, 79(4), 201–206. doi:10.4046/trd.2016.79.4.201
Future Hopes of Eradicating Tuberculosis
The World Health Organization estimates that one-fourth of the global population is infected with TB or LTBI with China, Russia, and India, making up half of those cases. Tuberculosis is still one of the top ten causes of death worldwide with 10 million new cases and 1.6 million deaths in 2017.
In the United States, there were 9,105 new cases of tuberculosis in 2017, but national rates have been in steady decline. In 1991 there were 10.4 cases per 100,000 in the U.S. and only 2.8 cases per 100,000 in 2017-a 73% difference.7,8 Eradication is dependent on early detection and treatment, and vaccination.6
Now Check Out the Peer-Reviewed, Self-Study CE Courses from Today’s RDH!
Listen to the Today’s RDH Dental Hygiene Podcast Below:
References
- American Dental Association. Tuberculosis. (n.d.). Retrieved from https://www.ada.org/en/member-center/oral-health-topics/tuberculosis-overview-and-dental-treatment-conside
- Centers for Disease Control and Prevention. Basic TB Facts | TB | CDC. (2016, March 20). Retrieved from https://www.cdc.gov/tb/topic/basics/default.htm
- Centers for Disease Control and Prevention. Guidelines for Infection Control in Dental Health-Care Settings – 2003. (2003, December 19). Retrieved from https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5217a1.htm
- Centers for Disease Control and Prevention. Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health-Care Settings, 2005. (2005, December 30). Retrieved from https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5417a1.htm
- Jain, P., Jain, I. (2014). Oral Manifestations of Tuberculosis: Step towards Early Diagnosis. Journal of clinical and diagnostic research: JCDR, 8(12), ZE18–ZE21. doi:10.7860/JCDR/2014/10080.5281
- Lee, S.H. (2016). Tuberculosis Infection and Latent Tuberculosis. Tuberculosis and respiratory diseases, 79(4), 201–206. doi:10.4046/trd.2016.79.4.201
- Sosa, L.E., Njie, G.J., Lobato, M.N., et al. Tuberculosis Screening, Testing, and Treatment of U.S. Health Care Personnel: Recommendations from the National Tuberculosis Controllers Association and CDC, 2019. MMWR Morm Mortal Wkly Rep 2019:68:439-443. DOI: http://dxdoi.org/10.15585/mmwr.mm6819a3
- World Health Organization. Tuberculosis (TB). (2018, September 18). Retrieved from https://www.who.int/en/news-room/fact-sheets/detail/tuberculosis