In late 2013 I was approached about writing an article to summarize some private independent research I completed, studying retipped dental instruments. This article was published by a dental hygiene industry journal and rocked the boat a little more than anticipated. At the time, I was working in private practice dentistry after finishing my Bachelor’s degree, where I learned how to conduct research so I thought I would give it a go. After all, what would be the harm in actually seeing if retipping was as bad as some of the industry circuit lecturers were painting it to be?
I was early in my writing career, and I thought hygienists needed to hear what I found. To get them to listen, I chose to title the article with a name the average clinical hygienist would want to pick-up and read. The original article entitled “Scary, Nasty, and Shocking” printed in November 2013. To follow up that article I have decided to outline some of the conversations that occurred following the print… so, here we go.
Hello Americans, this is Emily Boge. Stand-by for news.
The year was 2012. After attending a lecture at a major Midwestern dental conference where I had heard a reputable speaker talk of the dangers of retipping dental instruments, I had dinner with a colleague and was talking about what I had learned at the lecture. I admitted that I had not known instruments could be retipped. That was not something that was done at the practice where I had worked for the previous nine years. And, as many times happens in dentistry, I was not privy to information from outside my operatory. After dinner, I went back to the hotel room and did a search on retipping companies. Online, there were many companies that invited me to send my dull and broken instruments to them with the promise of replacing them with brand new, rejuvenated instruments for under two dollars an end.
I chose to investigate whether what the lecturer said was true, or if it was possible to obtain a new instrument from these companies at a fraction of the cost with the same, or similar, quality. I did not have funding for the study and, being a full-time graduate student, I certainly did not have money to pay anyone to help me. I collected a box of random instruments that had been retipped from colleagues to compare them to their direct-from-the-factory counterparts. I did not ask where they were retipped, or when, but just for some examples of retipped instruments. All of these instruments were labeled “RETIPPED” or “REFURBISHED,” as required by current law. I spent a few months studying the visual and tactile entities of these instruments; the cutting edges, the angles, the balance, everything geeks like me like to examine in dental instrumentation.
For those of you who did not read the original version of the article, here is a summary. When an instrument is retipped, the retipping facility removes the entire turning from the product shank and replaces a new turning in the used instrument handle using force and heat treating.
The results were pretty incredible. The original article gives the play by play and can be accessed here, but the emails and chatter online following the article were what really surprised me. I was not the only clinician who did not realize retipping existed, although one “commenter” on my article claimed I was fabricating my entire story about not realizing retipping actually happened. There were other clinicians like me, trust me- many of them emailed me!
I started receiving emails from colleagues across the country with stories of instruments with liquid trapped inside from the ultrasonic instrument baths or steam sterilization units, melted resin handles, instruments that were returned with indistinguishable terminal shanks, and many other deviations from “factory” quality. Other clinicians that were retipping thanked me for letting them know they were not the only ones seeing these concerning results.

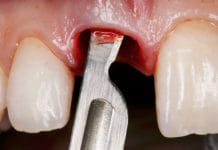
Picture 2: Blade angulation: The instrument on the right side shows a new 339 scaler. The left side instrument is a retipped 339 scaler.
Another layer of responses was the emails from dental instrument manufacturers thanking me for my candid article, written from a clinician’s standpoint. One manufacturer wrote to inform me of a product called Quik Tip™ Cone Sockets that was an environmentally friendly option to retipping. With Quik Tip™ Cone Sockets, clinicians could remove the small threaded tip of their dental instruments and replace them with a new tip that was factory-made to be replaced and tightened with a small manual wrench. These instruments were designed and engineered to be replaced in this manner, aligning cutting edges and angulations, while still reusing the handle and maintaining factory quality standards. Quik Tip™ Cone Sockets are an option to decrease the price for the spending-conscious consumer who desired savings. After researching this product, I decided the product may not be what I preferred at the time, but it was a really great alternative to retipping. Also, this product was available in the lighter resin handles, and the handles are not melted through the forcible retipping process.
Retipping companies were the final layer of response communications. On their websites, they teach the benefits of the retipping process, stating the cost savings and environmental friendliness of retipping are the main appeal to private practice dental offices. Some responses bordered on the lines of personal attacks, claiming I was under the payroll of dental instrument manufacturers (which at the time I was not) and I aimed to take away their ability to make a profit as a business. I still do not understand why it is legal to accept a medical device and permanently alter that device, returning it to the owner with the claim that it is back to its fully functional use.
All reputable domestic instrument manufacturers I have questioned about retipping have the same response; having a dental instrument retipped voids all warranty that involves the useful life of the instrument. These manufacturers care a great deal about the quality of the medical devices they produce, and that clinicians use, in the marketplace. Substandard methods of attempting to reproduce factory-quality, while still leaving the original factory name on the product, are a misrepresentation of these industry brands. Retippers are required to mark the instrument as “RETIPPED” or “REFURBISHED” per FDA guidelines but can leave the original manufacturer’s name on the product. Consistency and manufacturing quality are the backbones of instrument manufacturers, and the individual instrument factories go to great lengths to comply with ISO standards for quality control and FDA standards for dental instruments as medical devices.

In addition to the risks of compromising the quality of the instrument for patients, clinicians can also be affected by lesser-quality instruments that have been retipped. When the angulation of blades is not in proper design, ergonomic concerns can arise. The clinician may have to compromise clinical body positioning to adapt to the inconsistencies in weight, sharpness, and shape. One small change in an instrument could result in additional physical, psychological, or responsive strain to the patient and the clinician. Several of the emails I received from colleagues following my original article included stories of tips falling out, and liability risks elevating when retipped instruments broke in the mouths of patients.

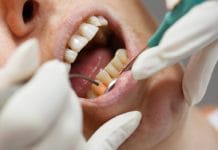
Picture 6: Poor quality of working end: The top instrument shows a retipped NEVI139 that had an invisible crack. The tip broke when adapted to a typodont. The bottome instrument is a new NEVI139.
As mentioned in my first article, the FDA has set standards for the use of dental instruments as medical devices, and states an instrument is a medical device because it “is intended to affect the structure or any function of the body of man or other animals, and which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of any of its primary intended purposes” (Food and Drug Administration, 1996). Dental professionals are responsible for taking every practical step in ensuring the maintenance, sharpening, handling, and sterilization of the dental instruments used to protect the safety of their patients. The safety and security of your patients and your own health as a clinician rest on you and the investment in quality dental instruments.
And now you know the rest of the story. Good day.
SEE ALSO: Ask Kara RDH: How Often Should I Retip My Instruments?
DON’T MISS: Science Shows Surprising Oral Health Benefits from Drinking Red Wine
Resource
- Food and Drug Administration (FDA). (1996). FDA Online. Medical devices: good manufacturing practice (CGMP) final rule: quality system regulation. Retrieved online at http://www.fda.gov/medicaldevices/deviceregulationandguidance/postmarketrequirements/qualitysystemsregulations/ucm230127